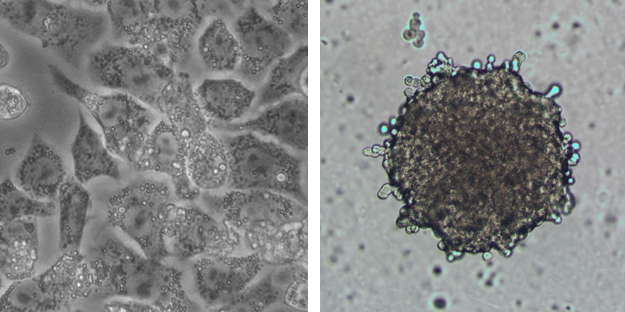
Drug discovery and development initiatives are increasingly moving from 2D to 3D cell culture models (e.g. spheroids, organoids, etc.) in order to better recapitulate the native tissue microenvironment and thus offer better predictive capacity of in vitro studies. However, when it comes to higher throughput screens in earlier stages of drug discovery and development, there exists some hesitation to make the move to 3D cell culture models, due to concerns that increases in model complexity will result in concomitant increases in cost.
However, depending on the cell model contents and the endpoints that are required, this is not necessarily the case. For example, if one considers 2D versus 3D culture of a cell line such as HepG2 cells, the perpetual nature of the cell line limits the costs to the price of the cell line itself. So, even though the seeding density of the 3D cell culture models is 10-fold lower, cells may be expanded in culture prior to seeding in 96 well plates for either 2D or 3D cell culture model generation, such that the cost of the cells is essentially the same (excluding minor costs incurred during expansion of cells). However, if one considers 2D versus 3D culture of a primary cell type like human hepatocytes, the single-use nature of these cells and high seeding density required by 2D culture results in nearly 70x lower per-well cost of cells for 3D vs 2D cell culture models!
| 2D Cell Culture | 3D Cell Culture | |||
|---|---|---|---|---|
| Cells per well | Cost per well | Cells per well | Cost per well | |
| Cell line | 1 x 104 | ≤ $4.53 | 1 x 103 | ≤ $4.53 |
| Primary cells | 7 x 104 | $17.08 | 1 x 103 | $0.26 |
*Cell line costs per-well are based on HepG2 cells from ATCC. Primary cell per-well costs are based on Gibco Metabolism Qualified Human Plateable Hepatocytes for 2D models or Corning 3D spheroid-qualified Primary Human Hepatocytes for 3D models. All calculations assume maximum use of cells within a vial from thaw (i.e. multiple plates would be seeded for most efficient use of cells). Per-well costs only include costs of cells.
It should be noted that this calculation is limited to evaluation of the cost of cells alone and does not account for the additional time and complexity introduced by some 3D cell culture approaches. For example, 2D cultures of primary human hepatocytes can be plated and ready for use in experiments in approximately 24 h, whereas 3D cultures require up to 7 days to enable complete aggregation of hepatocytes. Moreover, while many vendors are moving towards qualifying specific lots of primary human hepatocytes for spheroid formation, other cell subtypes may be less characterized and require evaluation of several lots of primary cell material and/or additional time in optimizing culture protocols to successfully generate 3D cell culture models for use in downstream assays.
Additionally, while many endpoints are easily translatable from 2D to 3D culture, assays on 3D cell culture models may require some optimization and/or additional assay time. For example, while 2D labeling is often quite straightforward, small molecule dyes and antibody labels can require optimization of permeabilizing protocols and additional incubation times to ensure complete penetration of these labels into 3D cell culture models. Moreover, analysis of such markers in an entire 3D model requires the acquisition of many more images (i.e. for a 200 µm spheroid, 20 separate images would need to be acquired if confocal imaging is used with a 10 µm z-step, whereas only a single image is required to capture cells in 2D cultures). However, this increased time can be bypassed in a higher throughput screening approach by only analyzing cells on the exterior of 3D models, such that only one image is required for analysis.
Interested in translating your cell culture models or assays from 2D to 3D, but not sure where to start? Reach out to info@visikol.com or contact us today for a complementary project consultation!