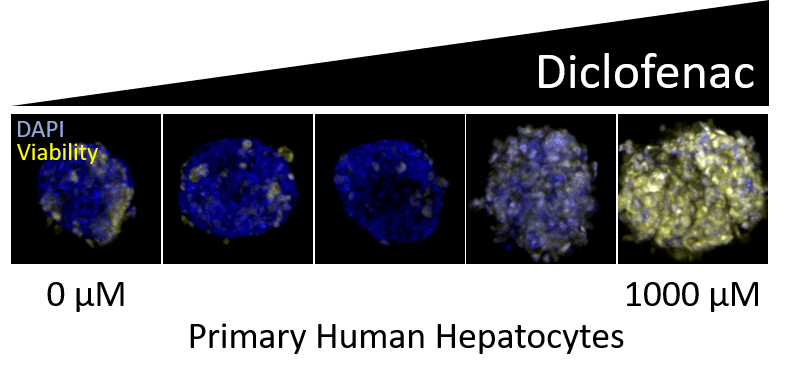
Drug-induced liver injury (DILI) is one of the most frequent causes of safety-related drug marketing withdrawal, according to the Food and Drug Administration (FDA).1 This makes models capable of evaluating hepatic toxicity an important aspect of the drug discovery process. At Visikol, we offer both 2D and 3D in vitro DILI models with specific emphasis on models capable of mimicking human liver functionality.
Human in vitro liver models typically include one of the following hepatocyte sources: HepG2, HepaRG or primary human hepatocytes (PHH). Each cell source has both benefits and downsides. HepG2 and HepaRG are both immortalized hepatic cell lines which are advantageous for their inter-assay consistency and proliferation ability, compared to primary human hepatocytes, which have limited proliferation capabilities and high donor-to-donor variability. However, PHHs are most similar to in vivo hepatocytes, being derived directly from human donors, and there are advantages to being able to evaluate toxicity across different types of donors, some who may be more susceptible to DILI.
Direct comparisons of these hepatocyte sources have been studied for use in toxicology evaluating both their gene expression and metabolic responses.2-4 As expected, there is often donor-to-donor variability in primary human hepatocyte assays.2-3 Base-line gene expression profiles have been shown to differ between all three types, but upon compound treatment showed more similar profiles.3 However, comparisons of metabolic cytochrome P450 capabilities and drug cytotoxicity showed greater similarity between PHHs and HepaRG, than HepG2.2-4 These studies demonstrate that determining the hepatocyte source that will be most relevant to your DILI questions is of great importance and we are here to help do just that.
Here at Visikol, we offer a variety of liver toxicity assays in both 2D and 3D model formats, which can include HepG2, HepaRG or primary human hepatocytes, additionally we are able to incorporate not only hepatocytes, but also non-parenchymal cell (NPC) types within our models, customizing to our Client’s interests and needs. These cellular models offer a selection of endpoints to examine DILI, including, but not limited to cell viability, apoptosis, reactive oxygen species generation, steatosis and phospholipidosis, cholestasis, fibrosis, mitochondrial function and glutathione depletion. If you are interested in drug-induced liver injury (DILI) models or would like to learn more about any of the other services, please reach out! We are always happy and excited to discuss our services and to learn more about your interests!
References:
- Food and Drug Administration. Guidance for Industry: Drug-Induced Liver Injury: Premarketing Clinical Evaluation (2009) http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM174090.pdf
- Gerets et al. Characterization of primary human hepatocytes, HepG2 cells, and HepaRG cells at the mRNA level and CYP activity in response to inducers and their predictivity for the detection of human hepatotoxins. (2012) Cell Biol Toxicol (28) p69–87
- Jetten et al. Baseline and genotoxic compound induced gene expression profiles in HepG2 and HepaRG compared to primary human hepatocytes. (2013) Toxicology in Vitro 27(7) p2031-2040
- Yokoyama et al. Comparison of drug metabolism and its related hepatotoxic effects in HepaRG, cryopreserved human hepatocytes, and HepG2 cell cultures. (2018) Pharm. Bull. 41(5) p722-732
