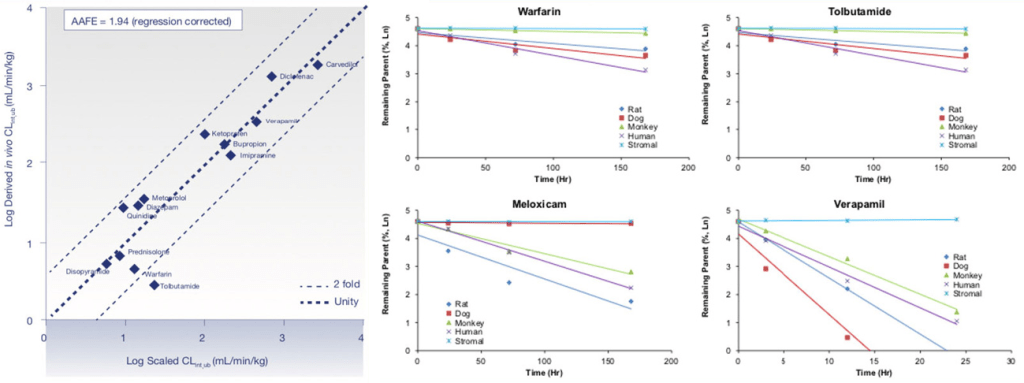
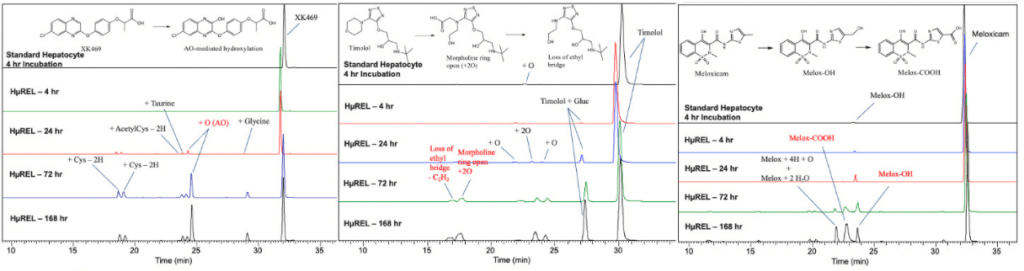
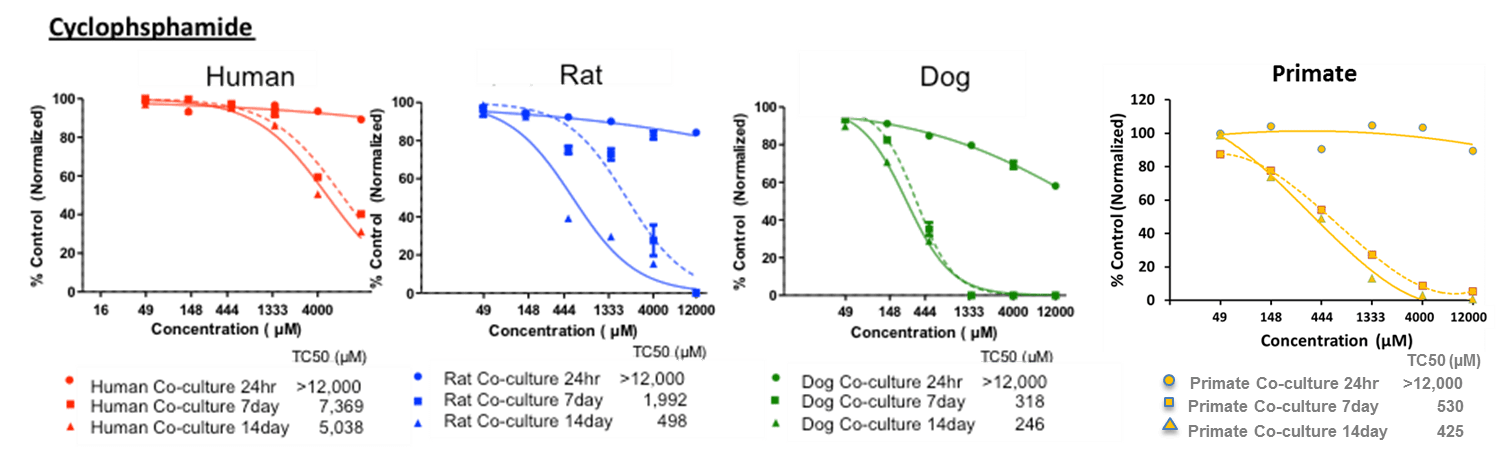
Services
Visikol leverages the HUREL micro liver models to provide its clients with a suite of best-in-class liver in vitro assays.
Multi-Parameter Toxicity
Our Multi-Parameter Toxicity assay’s apply HUREL’s superior metabolic competency to a panel of complementary assays, creating a multi-parametric characterization of hepatotoxic risk. HUREL Tox™ incorporates a 14-day, repeat-dose cytotoxicity study with both ATP and albumin (ELISA) readouts.
Reaction Phenotyping Assay
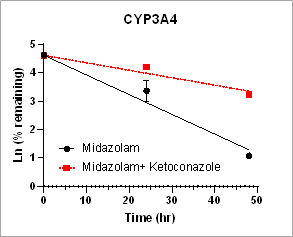
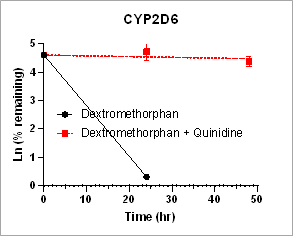
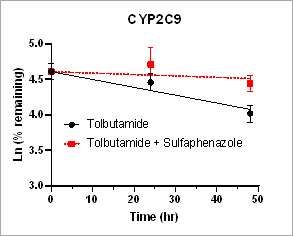
Using HURELhumanPool™ primary hepatocyte micro liver model, intrinsic clearance rates for parent drug depletion in the absence and the presence of CYP inhibitors are measured to determine the contribution of CYP enzymes. This information is useful to identify the specific enzymes responsible for the drug metabolism.