Overview
Detecting the presence of a virus in specific cells or in whole tissue sections is important to many researchers who wish to investigate the pathology of certain viral infections, examine the transfection efficiency of genetically modified viruses, or to identify the infection of cancer cells by a potential oncolytic viral candidate. The target tissue type, site of viral replication, and extent of infection are all important metrics that can be surveyed using immunolabeling along with 3D imaging of tissue sections. Researchers at the La Jolla Institute for Allergy & Immunology have recently published work conducted in part by Visikol [1].
Protocol
| Instrument | ThermoFisher CX7 LZR |
| Analysis Method | Whole Mount Imaging |
| Markers | Antibody(ies) for biomarker of interest (to be provided by customer or purchased by Visikol and billed through) Fluorescent Nuclear counterstain (e.g. DAPI) |
| Sample Submission | Whole Tissue fixed and stored in PBS with 0.05% azide, Formalin Fixed Paraffin Embedded (FFPE) tissue blocks, Tissues embedded with OCT, fixed 3D cell culture models in 96 or 384 well plate |
| Imaging Parameters | 4X, 10X, 20X, 40X magnification Step size (um) 5, 10, 25 (others available on request) |
| Image Analysis | Biomarker identification/localization Cell count positive for biomarker Morphological analysis for features such as shape, signal area, signal intensity (others available on request) |
| Data Delivery | Images in RGB format, ROI masks (e.g. positive cells), data tables containing cell counts and/or other feature data |
General Procedure
- Tissue sample is fixed and transferred to Visikol in PBS w/ 0.05% azide or in a form most appropriate for the customer (e.g. FFPE, OCT compound).
- The sample is processed and immunolabeled for biomarker(s) of choice and counterstained for DAPI.
- The sample is then cleared, mounted and imaged with High content imaging platform using desired parameters and objective magnification.
- The images are then processed and analyzed according to customer specifications.
- Images and quantification report are then transferred to the customer.
Data
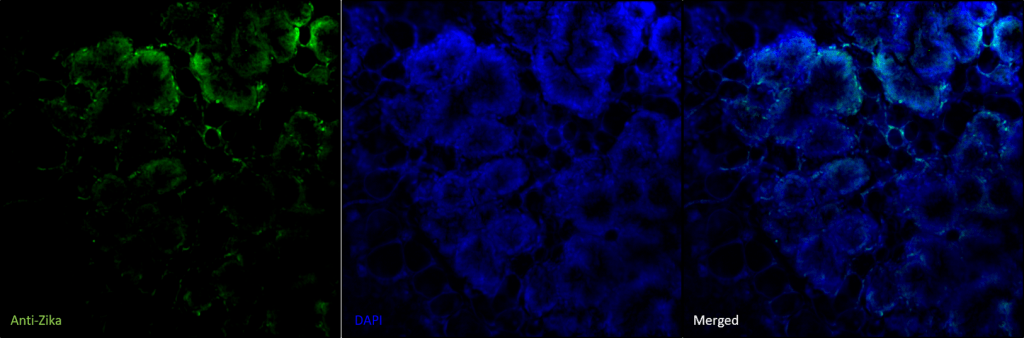
Figure 1. Identification and localization of Zika infection in mammary gland tissue.
References:
- Kim, K., Viramontes, K.M., Vozdolska, T., Huynh, A.-T., Villani, T., Gardner, G., Johnson, M., Ferro, P.J., Shresta, S., Nava, J.A.R. (2019). “Detection of Zika Virus in Mouse Mammary Gland and Breast Milk”. PLOS Neglected Tropical Diseases. in press.
