The brain is the central control system of our bodies, which is why it is so heavily protected by a robust barrier to keep us healthy and safe from harmful substances. While this protection mechanism is critical to brain health and overall wellbeing, it also proves to be a barrier to drug development. How do we treat neurological diseases if the brain has a fortress to keep out foreign articles – and further, how do we effectively assess the ability of a therapeutic to pass the blood brain barrier in vitro? Read more below to learn about how Visikol approaches this obstacle to drug discovery, and which features set our BBB assay apart.
 Our BBB Assay
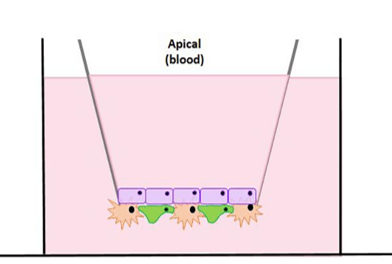
Our BBB Assay
Visikol’s BBB assay is unique in the sense that it is a triculture model, composed of iPSC-derived human brain epithelial cells, human astrocytes, and human pericytes, seeded around a transwell. The transwell serves to establish both an apical (or “blood”) compartment and a basal (or “brain”) compartment, in which your test articles can be dosed and collected to determine permeability across the BBB. This co-culture set-up not only mimics the characteristics of the BBB in vivo but also increases the expression of tight junction proteins, occludin and claudin. The cells in our model have been shown to express some of the key epithelial cell markers as well, such as BCRP, GLUT-1, MRP1, transferrin, P-gp, ZO-1, CD98hc, VE-cadherin, and CD31.
Static System vs. Flow System
You may be thinking to yourself, “how does a static system compare to a flow system?” Our answer is that while there are advantages and disadvantages to both systems – which are heavily dependent on the use case – Visikol’s BBB assay is higher throughput than a traditional flow system. Having validated the model in a 96-well format, our clients can assess more test conditions on a single plate, thereby receiving more comprehensive results in a cost-effective and timely manner.
 Visikol’s QA Measures
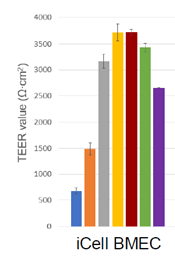
Visikol’s QA Measures
Another benefit to working with Visikol is that there are multiple QA measures in place to ensure the assay is operating as it should. Throughout the course of the assay, our scientists assess the trans-endothelial electrical resistance (TEER) values to ensure strong barrier integrity, including checks prior to running the assay and prior to collections. By assessing TEER values periodically, our scientists can ensure optimal assay performance and our clients can receive results they feel confident in.
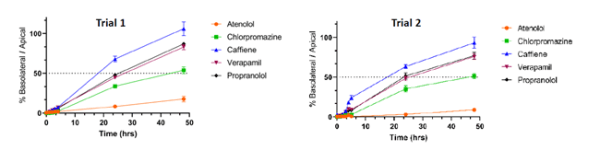
Reproducible Results
Lastly, one other element of Visikol’s BBB assay that sets it apart is its reproducibility. The model can differentiate between small molecule reference compounds with different mechanisms of transport through the BBB (passive diffusion, efflux, influx pumps), and it has demonstrated similar performance of these compounds in different trials, on different days.
If you are thinking Visikol’s BBB assay might be a solution for you and you would like to learn more, reach out to us to chat with our team: Get Started Today
