The tumor microenvironment (TME) is the ecosystem surrounding a tumor which includes the surrounding blood vessels, fibroblasts, immune cells, signaling molecules and the extracellular matrix (ECM). The tumor and its surrounding microenvironment are in constant interaction and influence each other, both positively and negatively. The TME gives a tumor all the nutrients it needs, promoting the survival and growth of the cancerous lesion. While some aspects of the TME are general across cancer types, populations of cells and the surrounding structures may vary for each individual tumor depending on cancer type, severity, and stage. Understanding the TME is becoming key in breaking down the development and progression of cancer to help understand and improve cancer treatment and precision medicine.
For example, many cancer types show a very fibrotic, or stiff, microenvironment, commonly promoted by the activation of cancer-associated fibroblasts (CAFs). This stiffer environment increases the difficulty for drugs to penetrate and reach the core of the tumor, promoting the need for adjustments in treatment. On the other hand, other cancer types tend to be more vascular or surrounded with dense blood vessels, as compared to the fibrotic TME, it may be easier for drugs to penetrate these tumors. The TME also contains many different immune cells and location of immune cells in respect to the tumor itself and other cell populations of the microenvironment. Understanding the localization of these cell populations as well as activation states and protein expression is important to evaluate treatments such as immunotherapy and checkpoint inhibitor treatments to develop new methods of treating cancer. The impact of the TME on every aspect of cancer and cancer treatment promotes the need for an improved method of visualizing and analyzing this microenvironment and techniques such as multiplex labeling opens the door to obtaining multitudes of information from a single specimen.
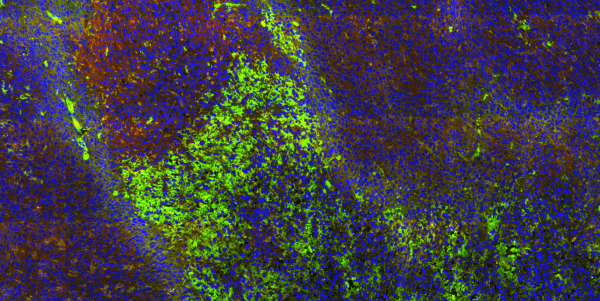
Tumor tissue fluorescently labeled with angiogenic protein VEGF (green), M2 macrophage marker CD206 (gold), leukocyte marker CD45 (orange), as well as cytotoxic marker CD8 (red), with nuclear counterstain DAPI (blue).
Multiplex Imaging
Visikol has developed a proprietary multiplex slide imaging approach, allowing visualization of over 10 markers per slide by conducting sequential rounds of labeling and imaging. Complex labeling using multiple rounds of immunofluorescence comes with a thorough optimization and validation for each antibody and panel prior to use in our platform. Recently, Visikol has optimized and validated a 5 panel, 19 marker, multiplex labeling approach to visualize and analyze the TME. The validated panel includes macrophage polarization (M1, M2a, M2b, M2c and M2d), CAF, and T-cell phenotypes, as well as a general TME profile (proliferation, apoptosis, hypoxia, and angiogenesis). These markers along with the image analysis offered at Visikol allow for an in depth look at cell populations, cell phenotypes, cell invasion into the tumor and spatial differences among the targeted cells. Together these techniques allow for the detailed evaluation and understanding of the TME as a whole ecosystem in cancer.
Validated Visikol TME Panel with Associated Markers
| Macrophage Polarization | CD68, CD80, CD86, CD163, CD206, VEGF |
| CAF Phenotype | αSMA, FAP, Vimentin, FSP1/S100A4 |
| T-Cell Phenotype | CD45, CD3, CD4, CD25, CD8 |
| TME Profile | Ki67, Caspase-3, VEGF, HIF1α, PanCK |
Visikol and its cutting-edge Multiplex technology and image analysis offers a new paradigm in oncology research and open a whole new door to understanding cancer development, progression, and treatment. If you’d like to learn more, visit our Multiplex Technology Page, our Overview Page, or our recent blog post about the Clinical Applications of Multiplex Imaging and how it can be utilized as a Clinical Tool. Reach out to our team today to if you want to learn more.
