
While most researchers think that the modern paradigm of tissue clearing and 3D imaging started with the CLARITY publication in 2013 at Stanford, it actually started before that at Rutgers University in a Plant Biology lab run by Dr. James Simon with the introduction of the Visikol® reagents. While working on his PhD in Medicinal Chemistry in Dr. Simon’s lab Dr. Tom Villani, the Chief Science Officer of Visikol, was using Chloral Hydrate to clear plant tissues when he thought up the idea for the Visikol® reagents. The original problem that Dr. Villani was trying to address was that while Chloral Hydrate was effective at rendering plant specimens transparent for microscopy, it was a DEA-controlled narcotic and thus challenging to obtain and to work with. Dr. Villani decided to instead develop a tissue clearing approach that was equally effective but was not a controlled narcotic, and thus the Visikol® reagents were developed and patented at Rutgers University. After a short period of time, Dr. Villani began to apply these reagents to animal tissues and thus the Visikol® HISTO™ reagents were developed, which have been used by over a thousand research labs from around-the-world to image their tissues in 3D. Over the last ten years, a lot has changed in this field and Visikol wanted to reflect on how far the field has come and what bottle necks may still exist.
What is Tissue Clearing?
Before we dive into the current state of the field, it’s important to give a refresher on what tissue clearing is and how it works. When you place your hand in front of your face, you of course cannot see through your hand even though your hand is mostly water (i.e., transparent) due to the fact that the lipids, protein and cytosol within your cells have different refractive indices. As light passes through each cell and these respective components, the light is diffracted and your hand appears opaque. This is the very reason why in life sciences we traditionally slice tissues into ultra-thin two slices, so that we can actually image and visualize them on a microscope. While this two-dimensional approach has been the traditional workflow for a century, the problem is that tissues are three-dimensional in nature and taking a cross section only samples a very small volume of the overall tissue.
Dr. Michael Johnson gives an overview of tissue clearing and how to employ tissue clearing to image tissues in 3D using confocal or light sheet microscopy.
Tissue clearing refers to manipulating a tissue using a chemical approach so that the refractive index across the entire tissue is relatively uniform, which makes the tissue optically transparent. This can be conducted using solvent based approaches (e.g., Visikol HISTO, BABB, 3DISCO, iDISCO, uDISCO), aqueous hyper-hydration techniques (e.g., ScaleS, FRUIT, TDE, ClearT, CUBIC) or hydrogel embedding techniques (e.g., PACT, PARS, CLARITY). Today, there are many techniques and dozens of variations of each for all sorts of applications. Each one of these techniques has its own unique advantages and disadvantages, and no single technique is best for all applications. For example, some approaches generate extremely transparent samples but can be incredibly challenging to execute and others might be rapid but and easy-to-use but not compatible with fluorescent protein, for example. Visikol has built a comprehensive overview of the varying techniques and their associated advantages and disadvantages: Tissue Clearing Comparison.
When these techniques are combined with fluorescent labeling and advanced microscopy (e.g., 2-photon, confocal, light sheet), they enable researchers to visualize tissues in 3D instead of traditional microscopy. A great example of this is the work that Visikol has done with Dr. Benedict Law’s Lab at Cornell on imaging whole brains or with Dr. Kacy Cullen’s lab at UPenn on Peripheral Nerve Repair Imaging. Tissue clearing allows these researchers to survey large volumes of data and to address research questions that require spatial context which is particularly useful in evaluating heterogenous tissues like the brain.
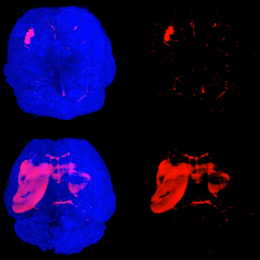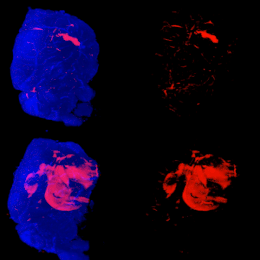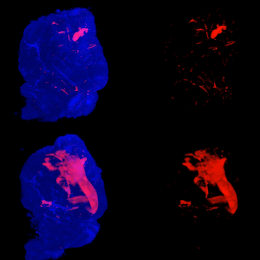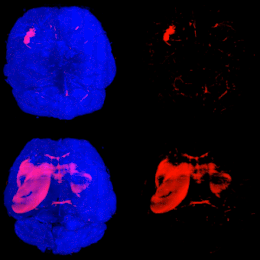
Mouse Brains Imaging using Light Sheet Microscopy and Tissue Clearing
Technology Advancements
When Visikol was first launched to commercialize tissue clearing reagents and kits, the space was very nascent and the techniques were used by only a select few researchers who were mostly focused on neuroscience. Therefore, much of the infrastructure required to image cleared tissues did not yet exist, and the microscopes, objectives, protocols and data handling approaches were primarily DIY which limited widespread adoption. However, over the last few years there are now several commercial light sheet microscope systems, several tissue clearing focused objectives and a multitude of protocols which ensure easier adoption. Furthermore, alongside many of the microscopes in the space are now software packages which can handle the immense amounts of data generated from these tissues. These advancements have combined to allow a wider community of researchers to now employ tissue clearing to image their tissues in 3D.
Dr. Michael Johnson gives an introduction to light sheet microscopes and how they can be used to image cleared and labeled tissues in 3D. Dr. Johnson gives an overview of how light sheet microscopes operate and the advantages and disadvantages with some common systems.
Different Tissue Clearing Techniques
Tissue Clearing with CLARITY
The CLARITY approach involves embedding a whole tissue in a polyacrylamide hydrogel and pulling an electrical current through the sample to remove the lipids from the cells. This process renders the tissue transparent through removing cellular components with a low refractive index while leaving the proteins, and nucleic acids intact within the hydrogel matrix.
CLARITY working in the brain.
BABB Tissue Clearing
BABB is an abbreviation for Benzyl Alcohol/ Benzyl Benzoate whereas the solution is comprised of two parts benzyl benzoate and one part benzyl alcohol. BABB has been used as an optical clearing agent for decades and works very well for many applications such as the clearing of bones. It is also compatible with immunolabeling and clears tissue quickly.
Tissue Clearing with SeeDB
Researchers have developed numerous aqueous clearing agents to preserve Fluorescent Protein and one of those clearing agents is SeeDB which is a combination of fructose (80.2% wt/wt) in water and α-thiogylcerol (0.5%) at a refractive index of 1.48. SeeDB is able to quickly render tissues transparent without causing swelling, but is not compatible with immunolabeling.
Tissue Clearing with 3DISCO/iDISCO
To clear myelinated tissues Erturk et al. developed 3DISCO which utilizes tetrahydrofuran (THF) instead of alcohol, dibenzyl ether (DBE) and sometimes dichloromethane (DCM) depending upon the tissue. Reiner et al. further developed the 3DISCO technique to improve immunolabeling and this process is referred to as iDISCO.
Limited Widespread Adoption
While the field has advanced and the barrier to adoption has been greatly reduced, one has to be honest with where the field is today and where it will likely go in the future. Visikol had hoped as researchers that tissue clearing would revolutionize the entire tissue imaging space and allow all researchers to add a third dimension to their research. Unfortunately, tissue clearing is by its nature an incredibly complicated process which involves an in-depth understanding of histology, immunolabeling, chemistry, advanced microscopy, complex image analysis and data processing. Therefore, its use in research will always be limited to questions which explicitly require spatial information and cannot be answered in a simpler and more inexpensive manner. Further, deploying tissue clearing requires a lab that is well-equipped with equipment as well as staff with disparate backgrounds that can work together to leverage the various disciplines required to implement a tissue clearing and 3D imaging workflow.
Client Applications
When Visikol had first envisioned applying tissue clearing to evaluating tissues, the most apparent application that we had thought of was using it to better diagnosis cancers, as cancer is spatially complex and heterogenous. While this seems obvious and that it would provide immense clinical value, the initial work has not been so clear. From an operational point of view, changing decades old histological workflows is quite challenging and the expense required to validate novel labeling, imaging and also image analysis approaches is too burdensome. Further, from a clinical point of view the interesting part about 3D tissue imaging is that while these approaches can greatly increase data, sensitivity, accuracy and the overall amount of tissue surveyed, these factors do not necessarily change or improve clinical treatments or outcomes.
The reason for this is that in many cases the current modalities for characterization are ‘good-enough’ and there are not a wide array of potential therapeutics from which a more sensitive characterization approach could better guide clinicians. Though many think that more data and more complexity are better, this has not been shown to be the case in practice and tissue clearing and 3D imaging will likely only be effective in the clinic where they can substantially change clinical outcomes, and current practices are greatly limited. For example, if a 3D imaging approach is 15% more sensitive than the current 2D manual approach for characterizing a tissue, this might translate into the exact same clinical treatment paradigms and ultimately the same outcomes. This would be mean that a histology lab would need to spend significant sums of money and revolutionize their workflows to achieve no patient or financial benefit which is not something that clinicians will be willing to do. Conversely, if there is a common misdiagnosis (e.g., false negative or false positive) that results in significant negative outcomes that can be alleviated through 3D imaging, then such an application might have clinical value.
Is tissue clearing right for your research? Check out Visikol’s 9 considerations to be sure.
3D renderings of multi-photon imaging sessions using a CLARITY lens, up to 2mm deep in intact barn owl tectal lobes (~3 x 6mm block) with autofluorescence in the green channel subtracted to produce high signal to noise. The label is microruby (rhodamine conjugated dextran) injected in vivo, and the owl was perfused with paraformaldehyde.
3D renderings of multi-photon imaging sessions using a CLARITY lens, up to 2mm deep in intact barn owl tectal lobes (~3 x 6mm block) with autofluorescence in the green channel subtracted to produce high signal to noise. The label is microruby (rhodamine conjugated dextran) injected in vivo, and the owl was perfused with paraformaldehyde.
The Future of Tissue Clearing
What Visikol has seen from its base of a thousand plus research labs using its products and 18 of the top 20 pharmaceutical companies using its services is that tissue clearing can find a place in routine research, both in academic and also industry settings. These applications tend to be in thinner tissues (500-1500 microns) where they can be labeled uniformly with antibodies and imaged with traditional confocal systems and not light sheet microscopes. There are many applications which can benefit from this type of imaging, but these same research labs still employ traditional 2D histological processing whereas tissue clearing is not a replacement for all imaging but instead just one more tool in the researchers’ tool box. Tissue clearing can also help researchers answer questions they otherwise could not answer, which in totality means that this platform is helping to accelerate the drug discovery and development pipeline. Over the coming years we foresee that the barrier of adoption to using tissue clearing will continue to be reduced, but the tool will still remain quite complex and only have utility in certain applications.
An isolated anterior prostate lobe (approximately 1 mm3) from an adult C57BL/6J mouse was fixed overnight in 4% paraformaldehyde and immunofluorescently stained with antibodies against E-cadherin (Cell Signaling Technology (3195S), Red) and Cd31 (BD Pharmingen (557355), Green). Shown is a 150 micron optical slice.
